Introduction
Variation in protein activity — even among proteins from the same batch — can be a significant nuisance in research and manufacturing. It is good practice to check protein quality after extended storage or when the source has changed, before committing to further experimentation. The quality control of biopharmaceuticals such as antibodies is especially critical in manufacturing settings, where process parameters including pH, temperature, CO₂, and cell culture metabolites all affect antibody quality.
Standard physicochemical characterization methods such as mass spectrometry are necessary but not sufficient — biological activity must be investigated as well. This has traditionally been done using endpoint assays such as Enzyme-Linked Immunosorbent Assays (ELISA). However, ELISA does not provide kinetic or affinity data. Surface Plasmon Resonance (SPR) offers a powerful alternative: it is label-free, enables real-time assessment of protein interactions, and requires minimal sample preparation.
Large SPR systems have been used for protein quality control, but access to centralized equipment is limited, samples must be pre-stored and transported, and consumable costs are high. Portable SPR devices, on the other hand, give scientists quick and easy access to the kinetic and affinity data needed to validate quality — without compromising sensitivity or specificity.
Quality Control with the P4SPR™
We demonstrate how easy it is to use the P4SPR™ to determine which source of anti-nucleocapsid antibody exhibits the best binding performance with the SARS-CoV-2 nucleocapsid recombinant protein. Four sources of anti-nucleocapsid antibody were introduced to a sensor surface immobilized with the SARS-CoV-2 nucleocapsid protein (Table 1), and the SPR response of each source was compared.
Download Application Note 5 — QC of Antibodies (PDF, 380 KB)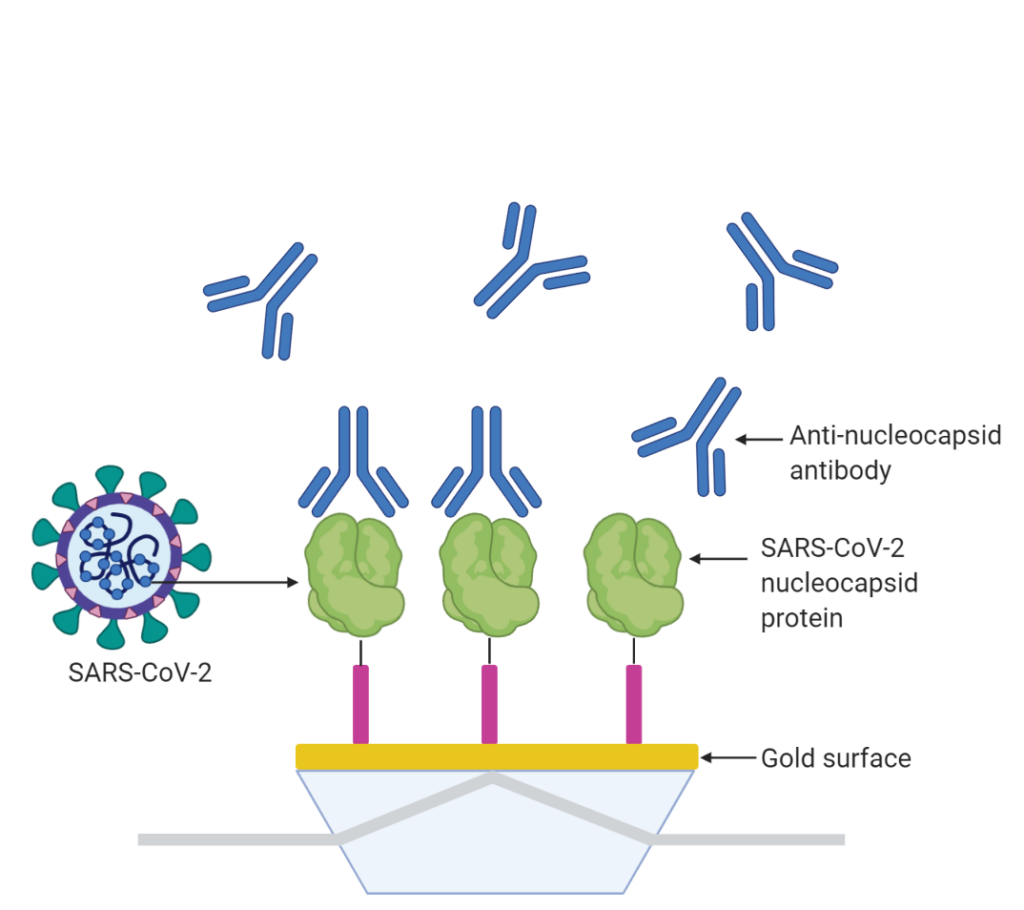
| Injection # | Antibody Source |
|---|---|
| 1 | AB1, lot A |
| 2 | AB1, lot B |
| 3 | AB2, lot A |
| 4 | AB1, lot C |
The four sources of anti-nucleocapsid antibodies yielded different average levels of binding, with the second source (Injection #2) giving the highest level of response. Notably, the entire experiment took approximately 2 hours — considerably shorter than a comparable ELISA assay.
Conclusions
A simple, portable SPR instrument such as the P4SPR™ can quickly establish which antibody source delivers superior binding performance, enabling researchers to disqualify less biologically active antibodies before committing them to further experiments. This reduces lost time and resources substantially.
The P4SPR™ provides a fast, real-time alternative to long and laborious endpoint assays for quality control of antibodies and other biopharmaceutical products. Its multichannel design allows multiple sources to be compared in a single experiment, and its ease of use means results can be obtained without specialized training or centralized lab access.
Contact us to learn more about SPR at info@affiniteinstruments.com.
Acknowledgements
We would like to thank Abdelhadi Djaileb from Dr. Jean-François Masson's lab at Université de Montréal for the collection of these data.
References
- M. Zschatzsch, Paul Ritter, Anja Henseleit, Klaus Wiehler, Sven Malik, Thomas Bley, Thomas Walther, Elke Boschke, "Monitoring bioactive and total antibody concentrations for continuous process control by surface plasmon resonance spectroscopy," Eng. Life Sci., vol. 19, pp. 681–690, 2019.
- P. Thillaivinayagalingam, J. Gommeaux, M. McLoughlin, D. Collins, A. R. Newcombe, "Biopharmaceutical production: Applications of surface plasmon resonance biosensors," J. Chromatogr. B, vol. 878, pp. 149–153, 2010.
- C. Gassner, F. Lipsmeier, P. Metzger, H. Beck, A. Schnueriger, J.T. Regula, J. Moelleken, "Development and validation of a novel SPR-based assay principle for bispecific molecules," J. Pharm. Biomed. Anal., vol. 102, pp. 144–149, 2015.
- Affinité Instruments. affiniteinstruments.com.