Every publication. One platform.
Browse the full list of peer-reviewed publications, book chapters, and preprints from labs worldwide — filterable by topic and searchable by keyword.
Browse publications →The lensless optical architecture isn't just what makes our instruments smaller. It's the cornerstone of the translational biosensing platform we're building.
How a patented optical foundation enables miniaturization, workflow integration, a path to automation, and signal quality you can trust — and why those are all consequences of the same design decision.
Read the technical note →Your Sensorgram Is Telling You Something.
We've traveled to labs on four continents. The most common thing we hear: "I'm not sure my data is good enough." Here's how to read what your sensorgram is actually saying.
Read the guide →Decoding SPR Sensorgrams
A visual reference for the most common sensorgram shapes — ideal binding, bulk RI shifts, baseline drift, non-specific binding, slow dissociation and more.
Open cheat sheet →Surface Chemistry Explained
Understand the two immobilization strategies — direct covalent coupling vs. capture-based — and which one fits your experiment.
Decoding SPR Sensorgrams
Quick visual reference for diagnosing common SPR data issues at a glance.
Static vs Kinetic SPR
When to use each mode, what data you get, and a side-by-side comparison table.
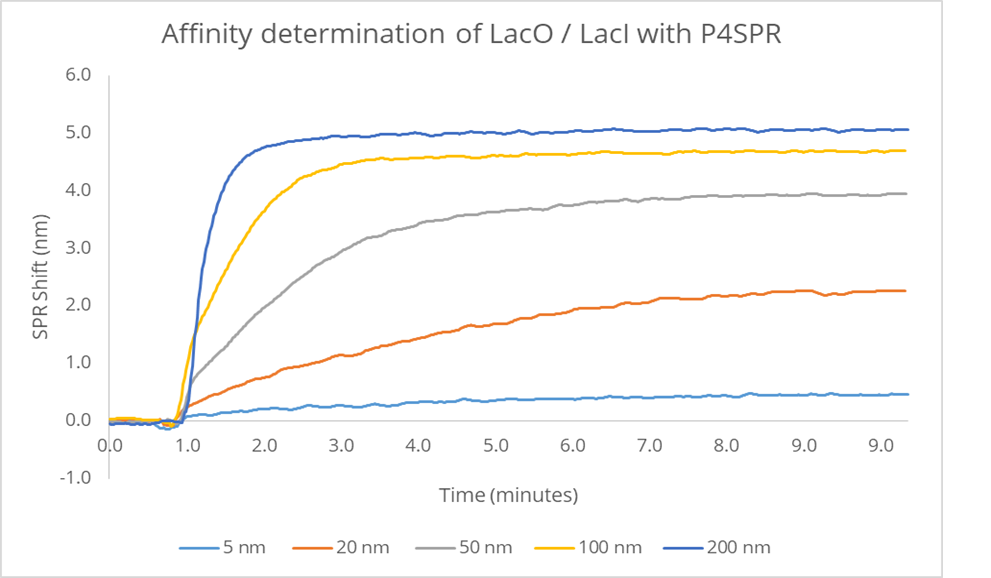
Gene Regulation — DNA-Protein Binding
Characterising the LacI–LacO interaction using the P4SPR in a label-free format.
Environmental Field Testing — RDX Detection
Real-world detection of explosive residues in water using portable SPR biosensing.
Protein–Small Molecule Binding
Pheromone–CD36 transmembrane protein binding characterised by SPR with sub-μM KD values.
Assay Optimization — Immunosensing
Oriented His-tag immobilization yields 5–10× greater surface density and superior reproducibility for anti-EcAII detection in patient serum.
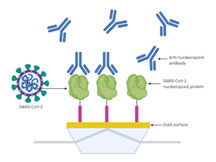
QC of Antibodies — SARS-CoV-2
Rapid label-free quality control of antibody preparations — activity and purity in one run.
Protein–Protein Interaction Screening
Rapid, label-free mapping of EntA–F biosynthetic pathway interactions using the P4SPR quad-channel platform.
P4PRO: Precision Binding Kinetics Made Simple
How Affinité's benchtop P4PRO delivers high-quality kon, koff, and KD data with a portable, user-friendly design.
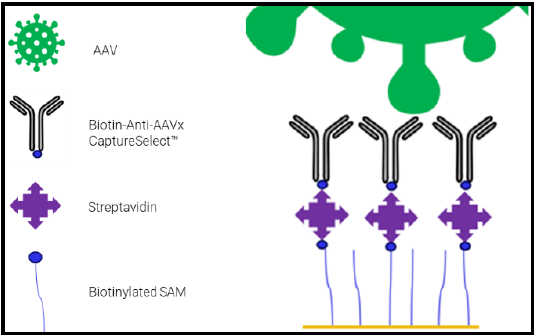
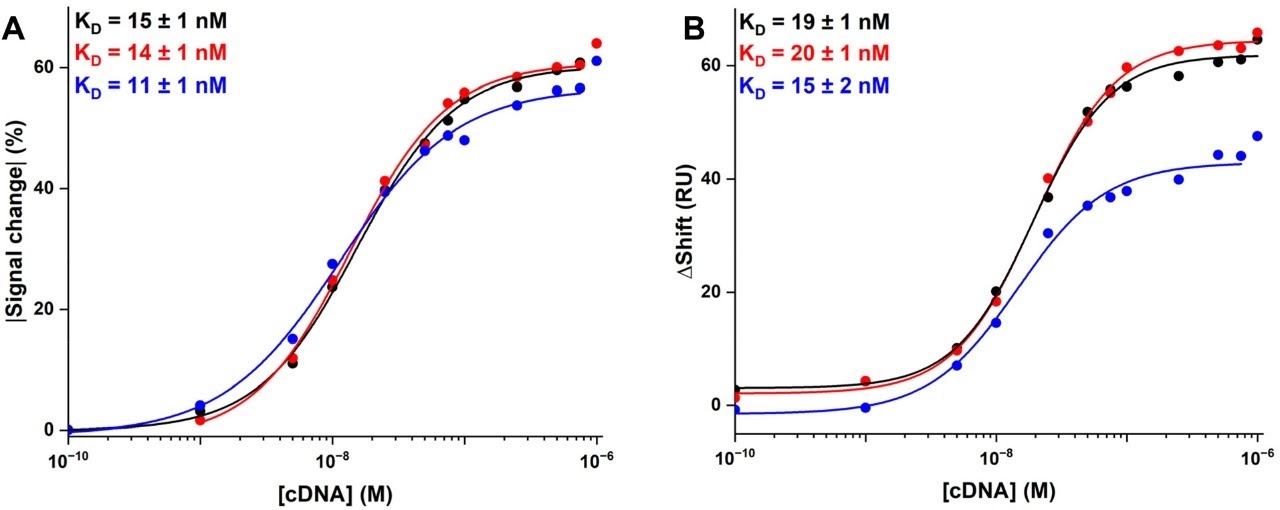
Monitoring AAV Bioproduction with SPR
Real-time SPR monitoring of AAV capsid binding during gene therapy vector production.
DNA Hybridization & Aptamer Biosensor Development with SPR
DNA probe screening with eSPR, split-aptamer hydrogels for vancomycin monitoring, PNA aptamer design for cardiac troponin I, and wearable translation.
Starter Test Kit: Immobilization & Binding Assay
Complete first experiment with the P4SPR 2.0 Starter Kit — EDC/NHS immobilization, 5-concentration IgG binding, KD fitting.
Covalent Immobilization of Proteins (EDC/NHS)
General-purpose EDC/NHS coupling on carboxyl sensors. Updated for AffiLabs.core v2.0.
Metal Chelation Immobilization (Ni-NTA / His-Tag)
Oriented His-tag capture via Ni-NTA for recombinant proteins. No activation or blocking required.
Streptavidin Capture of Proteins
High-affinity streptavidin–biotin capture for biotinylated molecules on SPR surfaces.
NaCl Calibration
Verify sensor sensitivity (≥ 1700 RIU/nm) using 5 NaCl concentrations. 30-minute procedure.
Static vs. Flow-Based SPR
Comparing static incubation and flow-controlled injection modes — when to use each and why.
The Four Key Concepts of SPR
SPR principle, instrument configuration, sensor classes (thin-film vs LSPR), and surface functionalization — the essentials for every user.
Manual Injection vs Pump-Assisted SPR
Steady-state vs. kinetic experimental setup — when to use each and what data you get.
SPR Sensor Surface Functionalization & Coupling Approaches
Sensor chip design, surface chemistry options (SAM, NTA, AffiCoat, streptavidin), and coupling method selection guide.
The SPR Sensorgram Explained
Five sensorgram phases, shape interpretation, quality features, and how the P4SPR generates binding data.
Issue 7 — Your Sensorgram Is Telling You Something.
Field notes from four continents, sensorgram diagnostics, and what we're building next.
Issue 6 — Your Instrument. Fully Unleashed.
AffiLabs.core is now available for the P4SPR — from run to results in one session, with SPARQ™ embedded.
Issue 5 — Lensless SPR, Better Data & a Transatlantic Adventure.
US patent granted, precision P4PRO kinetics case study, and the carry-on test.
Issue 4 — Looking Back on 2025.
Global lab visits, the three SPR questions we heard everywhere, and what we're building for 2026.
Issue 3 — Mobile SPR: Don't Wait for the Core Facility.
On-site SPR testing, 20+ conditions in 2 hours, and a kinetic vs. static explainer.
Issue 2 — Ten Years: mAb Case Study & Platform Evolution.
In-facility antibody characterization, product growth, and a look at mobile SPR.
Issue 1 — Affinité Turns 10: Small Instruments, Big Vision.
The founding story, 500+ researchers worldwide, and what a decade of lensless SPR looks like.

A Simple SPR Solution for Complex Samples
PEGS 2025 · Zhao S., Ricard P., Meredith R., Campbell C., Live L. — mAb characterization during biomanufacturing and IgG1–Protein A kinetics with the P4SPR.