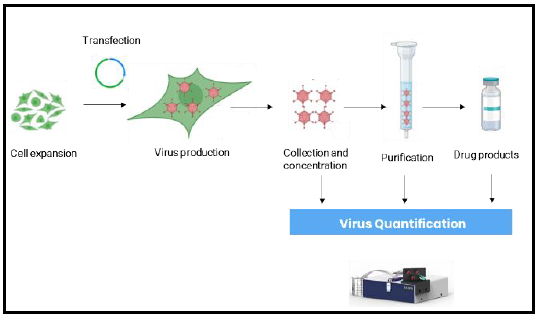
Introduction
Adeno-associated viruses (AAV) are increasingly produced for their potential in gene therapy, having shown promising results delivering therapeutic genes to target cells and tissues. As demand for AAV-based therapies grows, consistent quality control at scale becomes critical.
Conventional characterization methods — enzyme-linked immunosorbent assay (ELISA) and droplet digital PCR (ddPCR) — are widely used for viral genome count, total particle quantification, and purity assessment. However, ELISA in particular is time-consuming, often requiring days in centralized labs. This delay hinders timely quality assessment at different stages of the production process.
Surface plasmon resonance (SPR) offers a faster, simpler alternative. Affinité's lensless SPR technology enables label-free, real-time quantitation of total AAV capsids at the bench — without labels or extensive sample preparation. This application note demonstrates the use of P4SPR and P4PRO for AAV analysis across multiple stages of bioproduction, including the potential for in-line and at-line integration.
AAV Total Capsid Titer
Total capsid titer measurement is currently dominated by ELISA. SPR offers a more efficient and cost-effective alternative — no secondary antibody, no substrate for color development, and no wash steps between reagents.
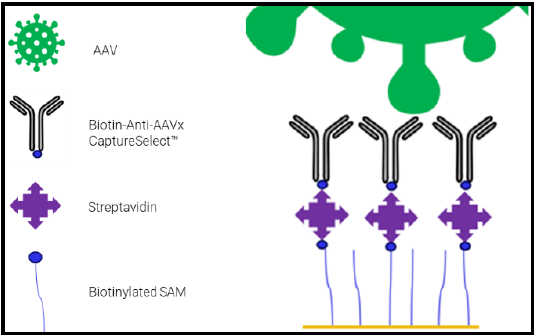
To produce different AAV serotypes, triple transfection was performed in HEK-293 cells adapted to serum-free media for suspension growth. Total capsid in purified samples was then analyzed by SPR. For titer analysis and surface regeneration, Affinité's biotin sensor and streptavidin kit were used to immobilize Biotin-Anti-AAVx CaptureSelect™ (Figure 2). Titer accuracy measured by SPR (in vg/mL) was compared to ddPCR, assuming one viral genome per particle. Results showed a correlation of 91–111% between the two methods.
Detection limit was determined to be in the 108 vg/mL range for both static and flow-based modes, using AAV2 at 1/1000 dilution.
Using purified AAV standards, calibration curves were established across multiple serotypes with R2 > 0.98 in all cases.
The 4-channel system provides one reference channel and three sample channels, enabling triplicate measurements that enhance data reliability and correct for bulk refractive index fluctuations. The SPR workflow has also been validated on crude in-process samples, enabling real-time monitoring at different stages of production without sending samples to a centralized QC lab.
In-Line and At-Line Integration
The compact footprint of the Affinité SPR instruments makes them suitable for use in BSL level 2 environments, without disrupting the BSL workflow or the SPR analysis. At-line testing reduces the need to access the QC lab — eliminating lengthy turnaround times and the risks associated with sample transport and handling.
In-line SPR can be installed directly in each viral vector clean room, delivering real-time results without removing samples from the controlled environment. This eliminates cross-contamination risk and enables timely production decisions based on on-site data.

P4SPR and P4PRO Advantages for AAV Bioproduction
Speed
Full titer experiment — 5 levels + regeneration — completed in under 2 hours vs. days for ELISA.
Label-Free
No secondary antibody or enzyme substrate required. Fewer reagents, fewer steps, lower cost per assay.
In-Process Samples
Validated on crude samples — monitor titer at each production stage without waiting for purified material.
Multi-Serotype
Calibration curves established for AAV2, AAV5, AAV9, phpEB, and CapB10 with R² > 0.98 across all.
BSL-Compatible
Compact enough for BSL 2 clean rooms. Enables in-line or at-line testing without workflow disruption.
Triplicate Precision
Three sample channels plus one reference channel deliver reproducible measurements with built-in drift correction.
Summary
SPR can quantify multiple AAV serotypes efficiently and reproducibly, offering a compelling alternative to ELISA for quality control in gene therapy bioproduction. The assay is fast, label-free, and has been validated on both purified standards and crude in-process samples. The Affinité P4SPR and P4PRO platforms support in-line and at-line integration in BSL 2 environments, enabling timely, on-site quality decisions throughout the AAV production pipeline.
Acknowledgements: Affinité Instruments thanks Dr. Marie-Eve Paquet and her team at CERVO, and Dr. Alina Sanchez from VVector Bio, for providing samples and valuable discussions.